A compromised paraventricular nucleus within a dysfunctional hypothalamus: A novel neuroinflammatory paradigm for ME/CFS
A neuroinflammatory paradigm is presented to help explain the pathophysiology of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). The hypothalamic paraventricular nucleus (PVN) is responsible for absorbing and processing multiple, incoming and convergent ‘stress’ signals, and if this cluster of neurons were affected (by neuroinflammation), the ongoing hypersensitivity of ME/CFS patients to a wide range of ‘stressors’ could be explained. Neuroinflammation that was chronic and fluctuating, as ‘inflammatory-marker’ studies support, could reflect a dynamic change in the hypothalamic PVN’s threshold for managing incoming ‘stress’ signals. This may not only be a mechanism underpinning the characteristic feature of ME/CFS, post-exertional malaise, and its associated debilitating relapses, but could also be responsible for mediating the long-term perpetuation of the disease. Triggers (sustained physiological ‘stressors’) of ME/CFS, such as a particular viral infection, toxin exposure, or a traumatic event, could also target the hypothalamic PVN, a potentially vulnerable site in the brains of ME/CFS susceptible people, and disruption of its complex neural circuitry could account for the onset of ME/CFS. In common with the different ‘endogenous factors’ identified in the early ‘neuroinflammatory’ stages of the ‘neurodegenerative’ diseases, an as yet, unidentified factor within the brains and central nervous system (CNS) of ME/CFS patients might induce both an initial and then sustained ‘neuroinflammatory’ response by its ‘innate immune system’. Positron emission tomography/magnetic resonance imaging has reinforced evidence of glial cell activation centred on the brain’s limbic system of ME/CFS patients. Neuroinflammation causing dysfunction of the limbic system and its hypothalamus together with a consequently disrupted autonomic nervous system could account for the diverse range of symptoms in ME/CFS relating, in particular to fatigue, mood, cognitive function, sleep, thermostatic control, gastrointestinal disturbance, and hypotension.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6291860/
Neuroinflammation in Patients with Chronic Fatigue Syndrome/Myalgic Encephalomyelitis: An 11C-(R)-PK11195 PET Study
Chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME) is a disease characterized by chronic, profound, disabling, and unexplained fatigue. Although it is hypothesized that brain inflammation is involved in the pathophysiology of CFS/ME, there is no direct evidence of neuroinflammation in patients with CFS/ME. Activation of microglia or astrocytes is related to neuroinflammation. 11C-(
R)-(2-chlorophenyl)-
N-methyl-
N-(1-methylpropyl)-3-isoquinoline-carboxamide (11C-(
R)-PK11195) is a ligand of PET for a translocator protein that is expressed by activated microglia or astrocytes. We used 11C-(
R)-PK11195 and PET to investigate the existence of neuroinflammation in CFS/ME patients. Methods: Nine CFS/ME patients and 10 healthy controls underwent 11C-(
R)-PK11195 PET and completed questionnaires about fatigue, fatigue sensation, cognitive impairments, pain, and depression. To measure the density of translocator protein, nondisplaceable binding potential (BPND) values were determined using linear graphical analysis with the cerebellum as a reference region. Results: The BPNDvalues of 11C-(
R)-PK11195 in the cingulate cortex, hippocampus, amygdala, thalamus, midbrain, and pons were 45%–199% higher in CFS/ME patients than in healthy controls. In CFS/ME patients, the BPND values of 11C-(
R)-PK11195 in the amygdala, thalamus, and midbrain positively correlated with cognitive impairment score, the BPNDvalues in the cingulate cortex and thalamus positively correlated with pain score, and the BPND value in the hippocampus positively correlated with depression score. Conclusion: Neuroinflammation is present in widespread brain areas in CFS/ME patients and was associated with the severity of neuropsychologic symptoms. Evaluation of neuroinflammation in CFS/ME patients may be essential for understanding the core pathophysiology and for developing objective diagnostic criteria and effective medical treatments.
http://jnm.snmjournals.org/content/55/6/945.long
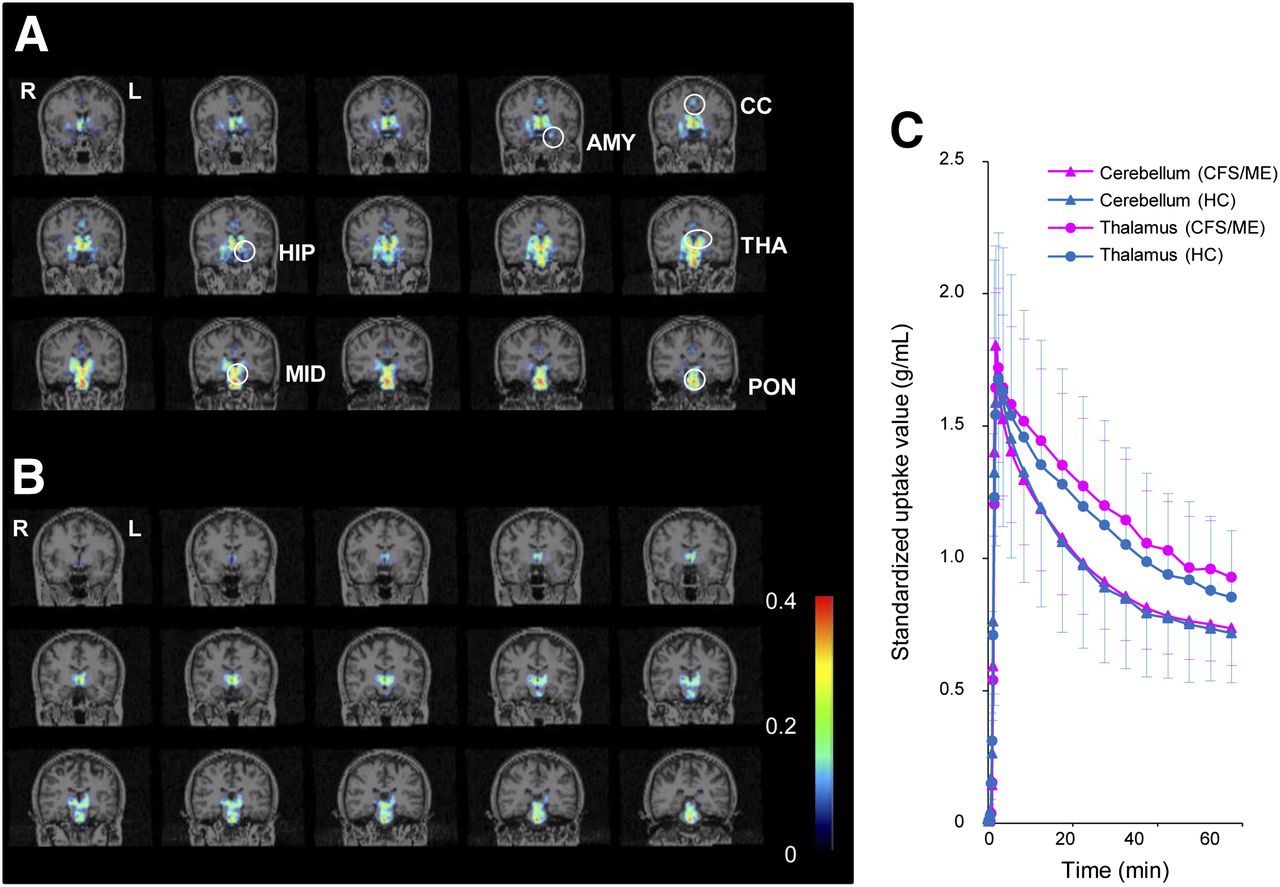
(A and B) Representative parametric PET images of 11C-(
R)-PK11195 binding in CFS/ME patient (A) and healthy control (B). Anatomic locations were mapped on coronal MR images. (C) Mean (±SD) regional tissue time–activity curves of 11C-(
R)(
R)-PK11195 for region of interest in cerebellum and thalamus in CFS/ME group and HC group. Scale indicates BPND. AMY = amygdala; CC = cingulate cortex; HC = healthy control; HIP = hippocampus; MID = midbrain; THA = thalamus; and PON = pons.
http://jnm.snmjournals.org/content/55/6/945/T2.expansion.html
@Murph @Hip