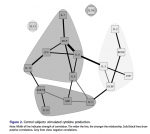
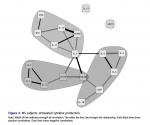
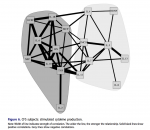
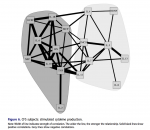
It occurs to me that it matters whether the PMBCs were tested in or out of the patients' serum.
I've read the methods section and I can't tell but maybe someone can:
Peripheral blood was drawn into lithium citrate tubes and processed within six to eight hours of sample procurement. All samples were collected and processed by the same clini- cal laboratory. Plasma samples were obtained from blood collection tubes which were spun for 20 minutes at 1000×g, and plasma aliquots were pipetted directly from each tube with phosphate buffered saline used for volume replacement. Afterward, heparinized peripheral blood was overlaid onto Ficoll/Hypaque and then centrifuged at 1000×g for 20 min. Cells at the interface were washed with Hank’s Balanced Salt Solution (HBSS) and enumerated with a standard hemocytometer.
PBMCs (1 × 106) were cultured in RPMI 1640 plus 10% fetal bovine serum (sup- plemented with glutamine, penicillin, streptomycin), with (stimulated) and without (constitutive) a mixture of phytohaemagglutinin (PHA) and phorbol-12-myristate- 13-acetate (PMA) (PMA @ 20 ng/well; PHA @ 0.05%/well) in 24 well plates for 48 hours at 370°C. After 48 hours, aliquots of supernatant fluid were collected and stored (–70°C) until analyzed. The following cytokines were evaluated using a mul- tiple analyte platform (BioPlex) and commercially available kits from BioRad (Bio- Plex Human Cytokine 17-plex): G-CSF, GM-CSF, IFN-γ, IL1β, IL2, IL4, IL5, IL6, IL7, IL8, IL10, IL12 (p70), IL13, IL17, MCP-1(MCAF), MIP-1β, and TNF-α. BDNF levels were determined using commercially available kits purchased from R&D Systems (Minneapolis, MN).