Ecoclimber
Senior Member
- Messages
- 1,011
J Virol. 2014 Feb 26. [Epub ahead of print]
Human Herpesvirus 6A infection in CD46 transgenic mice: viral persistence in the brain and increased production of proinflammatory chemokines via TLR9.
Reynaud JM1, Jégou JF, Welsch J, Horvat B.
Author information
Abstract
Human herpesvirus 6 (HHV-6) is widely spread in the human population and has been associated with several neuroinflammatory diseases, including multiple sclerosis.
To develop a small animal model of HHV-6 infection, we analyzed the susceptibility of several lines of transgenic mice, expressing human CD46, identified as a receptor for HHV-6.
We showed that HHV-6A (GS) infection results in the expression of viral transcripts in primary brain glial cultures from CD46-expressing mice, while HHV-6B (Z29) infection was inefficient. HHV-6A DNA persisted for up to 9 months in the brain of CD46-expressing mice but not in the non-transgenic littermates, whereas HHV-6B DNA levels decreased rapidly after infection in all mice.
Persistence in the brain was observed only with infectious but not heat-inactivated HHV-6A. Immunohistological studies revealed the presence of infiltrating lymphocytes and monocytes in periventricular areas of the brain of HHV-6A-infected mice.
Furthermore, HHV-6A stimulated the production of panel of proinflammatory chemokines in primary brain glial cultures, including CCL2, CCL5 and CXCL10 and induced the expression of CCL5 in the brains of HHV-6A-infected mice.
HHV-6A-induced production of chemokines in the primary glial cultures was dependent on the stimulation of toll-like receptor 9 (TLR9).
Finally, HHV-6A induced signaling through human TLR9 as well, extending thus observations from the murine model to human infection.
Altogether, this study presents a first murine model for HHV-6A-induced brain infection and suggests a role for TLR9 in the HHV-6A- initiated production of proinflammatory chemokines in the brain, thus opening novel perspectives for the study of virus-associated neuropathology.
IMPORTANCE SECTION:
Human herpesvirus 6 (HHV-6) infection has been related to neuroinflammatory diseases, however, the lack of suitable small animal infection model has considerably hampered further studies of HHV-6-induced neuropathogenesis.
In this study, we have characterized a new model for HHV-6 infection in mice expressing the human CD46 protein. Infection of CD46 transgenic mice with HHV-6A resulted in a long-term persistence of viral DNA in the brains of infected animals and was followed by lymphocyte infiltration and upregulation of the CCL5 chemokine, in the absence of clinical signs of disease.
The secretion of a panel of chemokines was increased after infection in primary murine brain glial cultures and the HHV-6-induced chemokine expression was inhibited when TLR9 signaling was blocked.
These results describe the first murine model for HHV-6A-induced brain infection and suggest the importance of TLR9 pathway in the HHV-6A-initiated neuroinflammation.
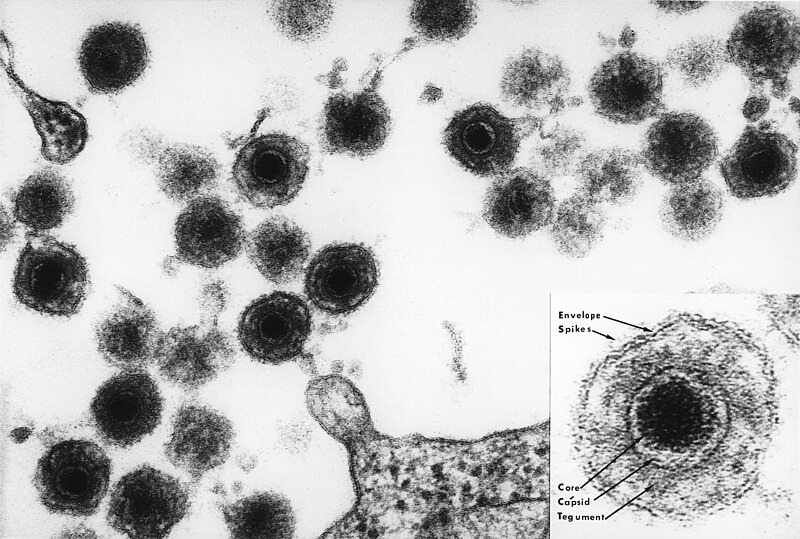
Human Herpesvirus 6A infection in CD46 transgenic mice: viral persistence in the brain and increased production of proinflammatory chemokines via TLR9.
Reynaud JM1, Jégou JF, Welsch J, Horvat B.
Author information
Abstract
Human herpesvirus 6 (HHV-6) is widely spread in the human population and has been associated with several neuroinflammatory diseases, including multiple sclerosis.
To develop a small animal model of HHV-6 infection, we analyzed the susceptibility of several lines of transgenic mice, expressing human CD46, identified as a receptor for HHV-6.
We showed that HHV-6A (GS) infection results in the expression of viral transcripts in primary brain glial cultures from CD46-expressing mice, while HHV-6B (Z29) infection was inefficient. HHV-6A DNA persisted for up to 9 months in the brain of CD46-expressing mice but not in the non-transgenic littermates, whereas HHV-6B DNA levels decreased rapidly after infection in all mice.
Persistence in the brain was observed only with infectious but not heat-inactivated HHV-6A. Immunohistological studies revealed the presence of infiltrating lymphocytes and monocytes in periventricular areas of the brain of HHV-6A-infected mice.
Furthermore, HHV-6A stimulated the production of panel of proinflammatory chemokines in primary brain glial cultures, including CCL2, CCL5 and CXCL10 and induced the expression of CCL5 in the brains of HHV-6A-infected mice.
HHV-6A-induced production of chemokines in the primary glial cultures was dependent on the stimulation of toll-like receptor 9 (TLR9).
Finally, HHV-6A induced signaling through human TLR9 as well, extending thus observations from the murine model to human infection.
Altogether, this study presents a first murine model for HHV-6A-induced brain infection and suggests a role for TLR9 in the HHV-6A- initiated production of proinflammatory chemokines in the brain, thus opening novel perspectives for the study of virus-associated neuropathology.
IMPORTANCE SECTION:
Human herpesvirus 6 (HHV-6) infection has been related to neuroinflammatory diseases, however, the lack of suitable small animal infection model has considerably hampered further studies of HHV-6-induced neuropathogenesis.
In this study, we have characterized a new model for HHV-6 infection in mice expressing the human CD46 protein. Infection of CD46 transgenic mice with HHV-6A resulted in a long-term persistence of viral DNA in the brains of infected animals and was followed by lymphocyte infiltration and upregulation of the CCL5 chemokine, in the absence of clinical signs of disease.
The secretion of a panel of chemokines was increased after infection in primary murine brain glial cultures and the HHV-6-induced chemokine expression was inhibited when TLR9 signaling was blocked.
These results describe the first murine model for HHV-6A-induced brain infection and suggest the importance of TLR9 pathway in the HHV-6A-initiated neuroinflammation.

Last edited:
