[35.03 to 41:46]
So, the other one... this is the brand new stuff that hasn't been discussed before and I'm really excited about this one because this is something that if it works, it will profoundly change how we think about ME/CFS and it will change how we treat it and it will open up a lot of interesting directions.
So, here's the basic idea.
We have a peripheral immune system and we have a central immune system. So in the brain the immune cells are called microglia and they're responsible for the whole immune defense of the brain and in our body we have T cells, B cells, natural killer cells - there's a lot of different cells in the body. Now those are supposed to be kept separate. So your peripheral immune cells - your T cells and B cells - they're not supposed to be in your brain and your microglia in your brain are not supposed to be in your body.
Now the blood brain barrier, which is a kind of a sheath that goes around all the vessels in your brain, it keeps those two things separate: it keeps the peripheral immune cells out of the brain because those peripheral immune cells are not trained to operate in the very sensitive environment of the brain. That's potentially dangerous if they're in there for too long, if they infiltrate the brain. So the blood brain barrier does a really good job of keeping those out.
There's a couple of different layers: there's these little tight endothelial cell junctions and then the astrocytes, which is some cells in the brain - they form these little lipid feet that form a different barrier. So there's a couple of barriers you have to get through to penetrate into the brain and in a healthy individual these things should always be kept separate.
Now what we're hypothesising, what's possibly happening in ME/CFS, is something has happened to this blood brain barrier.Either it's been damaged or for some reason the astrocytes are kind of opening the gate and what is happening - again this is just, we haven't shown this, this is just a hypothesis - is that these gates have opened and it's letting the peripheral immune cells squeeze through into the brain and then they start causing inflammation, which is making you feel fatigued and making you feel sick.
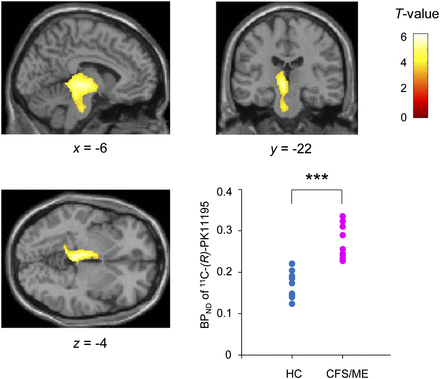
Now this picture right here is from multiple sclerosis and we think this also happens with multiple sclerosis but in MS, the cells that get through are trained to attack your own neurons and so that's a very devastating illness. In ME/CFS, we don't think autoimmune cells are getting through, we think other cells are getting through but they're enough to cause inflammation. They're not directly attacking your neurons but it's creating inflammation that's making you feel sick. So kind of a lesser form of multiple sclerosis would be one way to look at it. Not as damaging but still makes you feel really bad. So if that were to happen all these cells in the periphery are going to punch through and start to cause inflammation and there's many cell types - you got your B cells, your NK cells, your T cells, and these could be causing an inflammatory response in the brain.
So I think this is why you probably saw recently that rituximab which is a B cell depleter, why it may work in ME/CFS because if you can give rituximab and deplete the B cells in your body, there won't be as many B cells to cross into the brain to cause problems. That's just my guess as to how it works but it all fits together with the hypothesis.
So anyway, how do we do a brain scan to see if this is happening? Because if we can find these peripheral immune cells in the brains of people with ME/CFS, that's going to be a breakthrough because they're not supposed to be there. So that would be something where you would say "oh look, here's the immune cells, this is probably what's causing the problem, they're not supposed to be there".
So to do that, we have to go back to positon emission tomography. We cannot do this with MRI right now. We may be able to in the future, we're looking at that because it's cheaper but right now we're going to use PET.
So what we do here is we take a person with ME/CFS, or a healthy control, and we do a blood draw and we take that blood and separate out the leukocytes (and the leukocytes are basically all those immune cells I was talking about). Then you take those immune cells and you incubate them with the radiotracer and so those immune cells will 'eat' the radiotracer and so now they've got the tracer inside them and then we can measure where those cells go. So once they've been with that tracer for about an hour, they eat it up, they cells are tagged, now we inject the person's immune cells back into their body, except now - they're regular immune cells, they still work, they just have a tracer inside them now - so now we can follow them.
Now what we'd do is we'll inject these cells back and then we'll bring the person back 24 hours later to scan their brain to see if any of those cells actually punch through into the brain and then we'll bring them back 3 days later to see what it looks like.
What we expect to see is that in the healthy brain no signal will be detected at all. There'll be no evidence of infiltration of the brain but we think in the ME/CFS individuals, we'll pick up that signal in the brain.
Now, again, what's really cool about the scan is that there's no background signal so if the cells don't get to the brain there will be nothing there, it'll just be total black. Other scans, there's a lot of noise: this, there's no chance of noise. So if there's no cells there, you're just going to get a black picture very easy to interpret. If there are cells there, you'll get a signal, it'll be kind of glowing on the screen and so you can say 'yes' versus 'no', it's as simple as that: "Yes, the immune cells made it to the brain" versus "No, they didn't".
So if we find consistently that the ME/CFS individuals have these cells in the brain but the healthy controls don't, that's going to be a major advance because then we know we can target central inflammation or we need to target those immune cells in the periphery before they have a chance to cross the blood brain barrier. Or maybe we need to try to give agents to repair the blood brain barrier. It opens up a lot of targets for treatments. So, we hope that works, we don't know, we have not even run the first participant. This is very, very complex stuff. It'll probably take us about 5 - 6 months before we're truly running participants. I'm working with Suzanne Lappy who's an excellent radiochemist here at UAB. So we're going to get there but it will probably be later this year before we're actually running this full steam but, you know, it's the kind of thing again where if the hypothesis is supported, it's going to be very clear to interpret and it's going to really change how we approach CFS.