@Janet Dafoe (Rose49)
Unfortunately i did not have any feedback regarding all of my previous posts from Ron. But please ask from him to read the following and give this Hypothesis a chance.If he believes that Liver Disease is out of the question and will not be considered by the OMF, please do let us know.
I will also link this post to Post-Accutane and Post-Finasteride Syndrome websites for reasons that -hopefully- will be understood later on .
I am also CCing the following people :
@Jesse2233 @Gondwanaland @adreno @Isaiah 58:11 @nandixon @JaimeS @Learner1 @znahle @guitarman01 @xptriado @cfs6691 @tatt @Hip @A.B. @Gondwanaland
Please look at the following screenshots :
Please note : We have a bed-bound patient saying that any medication makes them extremely ill. We
know that the Liver processes Medications.
Whenever i talk with any ME/CFS Patient i always try to find where the Liver is involved. So the dialog proceeds as follows :
We now move on to the following screenshot i took today. The User
@cfs6691 asks
@Janet Dafoe (Rose49) :
and more mentions on Liver/Fibroscan/Liver Fibrosis on Phoenix Rising :
http://forums.phoenixrising.me/inde...rview-with-dr-julia-newton.23320/#post-455864
http://forums.phoenixrising.me/index.php?threads/dr-says-lyme-disease-does-not-exist.36648/page-7#post-643983
and one more :
We have mentions about problematic Fibroscans/Liver Fibrosis findings in PR. No action taken so far for further investigation (i also have Fibroscans showing Liver fibrosis that i could send to Dr Davis or anyone else interested ) :
Note that we are not talking about NAFLD (which is common) but Liver Fibrosis.
We move on to Accutane and Finasteride
http://forums.phoenixrising.me/inde...-it-to-this-me-chronic-fatigue-illness.54073/
http://forums.phoenixrising.me/inde...-58-70-response-rate.20430/page-5#post-889100
http://forums.phoenixrising.me/index.php?threads/propecia-finasteride-and-progesterone.34424/
http://forums.phoenixrising.me/inde...t-thyroid-resistance.11593/page-4#post-613272
http://forums.phoenixrising.me/index.php?threads/i-feel-like-giving-up-on-life.50299/
I now move on to the latest findings in hope that someone will listen or a Researcher may find something useful.
@Jesse2233 Please forward to your Researcher friend.
======Beginning of Post===============
Original post :
http://bit.ly/2gqDLCt
Before continuing, the reader is reminded that the Network Analysis referenced in the posts has been generated on April 2017. For a number of posts we will be referring to this version of Network Analysis.
However, the latest Network analysis and algorithmic runs (not shown at present) have been suggesting the potential importance of Glucokinase.
From the Wikipedia entry about Glucokinase, we read :
"
Most of the glucokinase in a mammal is found in the liver, and glucokinase provides approximately 95% of the hexokinase activity in hepatocytes. Phosphorylation of glucose to glucose-6-phosphate (G6P) by glucokinase is the first step of both glycogen synthesis and glycolysis in the liver."
Noticing the mention about Glucose-6-Phosphate, we read from the Wikipedia entry of Glucose-6-Phosphate :
"Within a cell, glucose 6-phosphate is produced by phosphorylation of glucose on the sixth carbon. This is catalyzed by the enzyme hexokinase in most cells, and,
in higher animals, glucokinase in certain cells, most notably liver cells. One molecule of ATP is consumed in this reaction."
In other words, Liver cells are responsible for the use of Glucokinase for phosphorylation of Glucose to Glucose-6-Phosphate. Then Glucose-6-phosphatase completes the final step of Glycolysis :
"
Glucose 6-phosphatase is an enzyme that hydrolyzes glucose-6-phosphate, resulting in the creation of a phosphate group and free glucose. Glucose is then exported from the cell via glucose transporter membrane proteins.
This catalysis completes the final step in gluconeogenesis and glycogenolysis and therefore plays a key role in the homeostatic regulation of blood glucose levels."
Given the above information, It is time to look at several new nodes as these were generated during the Network Analysis (April 2017) :
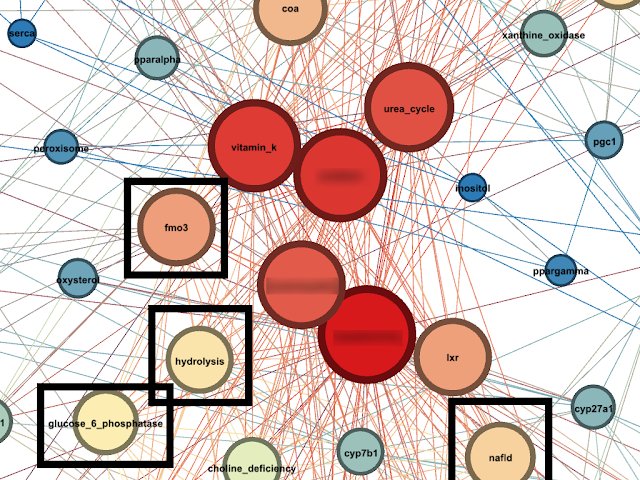
Recall that Glucose-6-Phosphate
hydrolyses Glucose-6-Phosphatase. We hypothesise that the Node named
Hydrolysis signifies this association. We note that
Hydrolysis is located next to
Glucose-6-Phosphatase node.
We also take note of a Node named
NAFLD (Non-Alcoholic Fatty Liver Disease).
Of particular interest is that Biotin and Retinoic Acid induce Glucokinase activity [1].
Furthermore LXR Receptor increases Glucokinase activity. In [2] we read (LGK= Liver Glucokinase):
"
In the current studies, we demonstrated that LXRalpha increased LGK expression in primary hepatocytes and that there is a functional LXR response element in the LGK gene promoter as shown by electrophoretic mobility shift and chromatin precipitation assay. In addition, our studies demonstrate that LXRalpha and insulin activation of the LGK gene promoter occurs through a multifaceted indirect mechanism"
Furthermore in [7] we read :
"Liver X receptor is a ligand-activated transcription factor, which is mainly involved in cholesterol homeostasis, bile acid and triglycerides metabolism,
and, as recently discovered, in the glucose metabolism by direct regulation of liver glucokinase."
We now turn our attention to NAFLD
: In [3] we read about the potential role of Glucokinase Regulatory Protein (rs780094 C>T ) to Liver Fibrosis severity of individuals having NAFLD :
Whether there is a higher prevalence of NAFLD and/or Liver Fibrosis in patients of the Syndromes discussed is currently unknown and should possibly be investigated.
We now look at FMO3 Node. In [4] we read :
"The FMO3 gene provides instructions for making an enzyme that is part of a larger enzyme family called flavin-containing monooxygenases (FMOs).
These enzymes break down compounds that contain nitrogen, sulfur, or phosphorus. T
he FMO3 enzyme, which is made chiefly in the liver, is responsible for breaking down nitrogen-containing compounds derived from the diet. One of these compounds is trimethylamine, which is the molecule that gives fish their fishy smell. Trimethylamine is produced as bacteria in the intestine help digest certain proteins obtained from eggs, liver, legumes (such as soybeans and peas), certain kinds of fish, and other foods.
The FMO3 enzyme normally converts fishy-smelling trimethylamine into another compound, trimethylamine-N-oxide, which has no odor. Trimethylamine-N-oxide is then excreted from the body in urine."
However, FMO3 has more functions, including Endoplasmic Reticulum Stress, LXR Regulation, Inflammation and Reverse Cholesterol Transport [4]
"
Knockdown of FMO3 in cholesterol-fed mice alters biliary lipid secretion, blunts intestinal cholesterol absorption, and limits the production of hepatic oxysterols and cholesteryl esters. Furthermore,
FMO3 knockdown stimulates basal and liver X receptor (LXR)-stimulated macrophage RCT, thereby improving cholesterol balance. Conversely, FMO3 knockdown exacerbates hepatic ER stress and inflammation in part by decreasing hepatic oxysterol levels and subsequent LXR activation. FMO3 is thus identified as a central integrator of hepatic cholesterol and triacylglycerol metabolism, inflammation, and ER stress. These studies suggest that the gut microbiota-driven TMA/FMO3/TMAO pathway is a key regulator of lipid metabolism and inflammation."
and :
"
Collectively, these results suggest that FMO3 knockdown promotes hepatic inflammation and ER stress in part by diminishing LXR activity. Collectively, our results are consistent with hepatic FMO3 serving as a critical determinant of the well-known ability of LXR to reciprocally regulate lipid metabolism and inflammation"
We also note the connection of Gut Microbiota to FMO3. The importance of LXR and Oxysterols has been discussed in previous posts.
In an attempt to connect the above with the Syndromes discussed, we find the following in the Wikipedia entry of Glucokinase :
"The mechanisms by which insulin induces glucokinase may involve both of the major intracellular pathways of insulin action, the extracellular signal-regulated kinase (ERK 1/2) cascade, and the phos-phoinositide 3-kinase (PI3-K) cascade.
The latter may operate via the FOXO1 transactivator."
In Wikipedia, we read :
"Forkhead box protein O1 (FOXO1) also known as forkhead in rhabdomyosarcoma is a protein that in humans is encoded by the FOXO1 gene.
FOXO1 is a transcription factor that plays important roles in regulation of gluconeogenesis and glycogenolysis by insulin signaling, and is also central to the decision for a preadipocyte to commit to adipogenesis.
It is primarily regulated through phosphorylation on multiple residues; its transcriptional activity is dependent on its phosphorylation state."
In [5] we find a very interesting discussion on the associations between Isotretinoin treatment, FOXO1 and :
-LXR Receptor
-Androgen Receptor (AR)
-DHT (Dihydrotestosterone which is blocked by Finasteride)
-5AR (5-alpha reductase which is blocked by Finasteride)
Some interesting excerpts :
"These data imply that isotretinoin treatment may downregulate the transcriptional activity of AR by increasing the nuclear concentration of the AR cosuppressor FoxO1. Furthermore, the isotretinoin-induced decrease of IGF-1 serum levels may impair IGF-1/PI3K/Akt-mediated nuclear export of FoxO1.
Moreover, IGF-1 is regarded as an androgen-dependent stimulator of 5α-reductase activity. In fact, experimental evidence has been provided for decreased androgen 5α-reduction in skin and liver of men with severe acne after oral isotretnoin treatment."
"
Furthermore, FoxO1 suppresses SREBP-1c, the key transcription factor of lipogenesis, in skeletal muscle by disrupting the RXRα/LXR heterodimer on the SREBP-1c promoter (Fig. 2C).219
Mice over-expressing FoxO1 lose their glycemic control and display a lower capacity for physical exercise due to severe muscle loss. Due to clinical experience, isotretinoin is administered with caution to athlethes who have to rely on their muscle mass and muscle strength."
In [6] we find an interesting case of individuals who received Isotretinoin and claim that they acquired Trimethylaminuria (which is directly associated with impaired FMO3 functioning) :
"
We want to raise awareness about the experiences of three users of Guía Metabólica suffering fish odor syndrome who underwent treatment with isotretinoin."
and
"Oral isotretinoin, a systemic retinoid, is often prescribed for the treatment of resistant acne. Its secondary effects are well known, particularly those related to bowel disease and the potentially teratogenic properties in pregnant women. Moreover, patients under treatment should undergo periodical transaminase analysis.
In fact, transaminase increases are described in 15-20% of patients, pinpointing the potentially hepatotoxic effects of isotretinoin. However, as far as we know, in the metabolism of isotretinoin there is no participation by FMO3."
and :
"
www.guiametabolica.org users suffering fish odor were treated with retinoic acid for 1 to 4 years with no other relevant side effects. None of them underwent urinalysis to measure trimethylamine, and none of their attending dermatologists suspected an acquired trimethylaminuria. Moreover, doing a search through other open support groups on the Internet we found at least two other adults reporting sustained fish odor after isotretinoin treatment."
References
[1] :
https://en.wikipedia.org/wiki/Glucokinase
[2] :
https://www.ncbi.nlm.nih.gov/pubmed/19366697
[3] :
http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0087523
[4] :
https://www.ncbi.nlm.nih.gov/pubmed/25600868
[5] :
https://www.ncbi.nlm.nih.gov/pubmed/22110774
[6] :
http://raredisorders.imedpub.com/isotretinoin-trimethylaminuria-and-theinternet.php?aid=8118
[7] :
https://www.ncbi.nlm.nih.gov/pubmed/25869557

